Audio and Web Conference
Video | Safely Maximizing COVID-19 Vaccine Dosing–Clarifying Syringes and More
View a recording of our March 17, 2021, live-streamed lab webcast. At the end of this webinar, attendees will be able to:
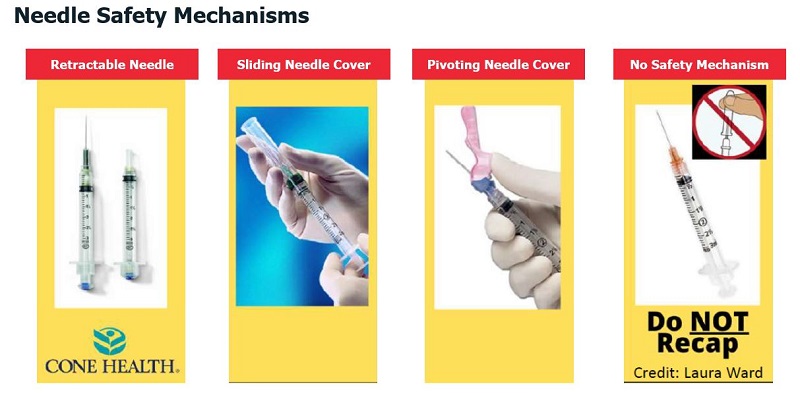
- Adhere to key steps for safe and efficient syringe handling during vaccine preparation and administration
- Understand information about error reporting
- Prepare for changes in the upcoming months in terms of necessary supplies
The presentation concludes with an informative Q&A session based on questions from hospitals around the globe.
Speakers
- Dr. Andrew Furman, Executive Director, Clinical Excellence, ECRI
- Michael R. Cohen, RPh, MS, ScD (hon.), DPS (hon.), FASHP, President, ISMP
- Kevin N. Hansen, PharmD, MS, BCPS, Assistant Director of Pharmacy, Moses H. Cone Memorial Hospital
- Jason Launders, Director of Operations, Device Evaluation, ECRI
- Kristina Cybularz, Manager, Supply Guide—Supply Chain Services, ECRI
- Juuso Leinonen, Senior Project Officer, Device Evaluation, ECRI
Lab tour recording
Supplementary materials
Reported COVID-19 vaccine errors
The Institute for Safe Medication Practices (ISMP) encourages facilities to report vaccine errors to its voluntary error reporting system. Reported errors include the following:
- Dilution errors leading to under- or overdose of vaccine
- Mixing errors with two-component vaccines (diluent instead of vaccine or wrong diluent such as sterile water)
- Air injected into vial instead of diluent
- Storage issues (unsegregated vaccine brands in refrigerator)
- Wrong vaccine given for dose 2 (not checking/documenting in immunization information system)
- Administration to wrong age group
- Waste of vaccine and not taking advantage of over-fill in vaccine vials
- Errors in scheduling second dose
- Look-alike vials (vaccine-monoclonal antibody mix-up)
- Shoulder injury related to vaccine administration (SIRVA)
Preventing errors with COVID-19 vaccines
- Verify competency of preparers and vaccinators (many are volunteers)
- Dispense pharmacy-prepared and labeled syringes when possible, or one person prepares and administers
- For mass vaccination, utilize a standard, organized process with independent double checks
- Maximize doses withdrawn from vials
- Separate vaccines in storage
- Plan for leftover vaccine
- Be prepared for allergic reactions
- Report vaccine errors and adverse reactions (U.S. requires reporting to VAERS); additional reporting to ISMP is voluntary
- Utilize immunization information systems
Additional resources
American Society of Health-System Pharmacists, Institute For Safe Medication Practices, U.S. Pharmacopeia: FAQ for Optimizing COVID-19 Vaccine Preparation and Safety
ECRI is here to help
Our interdisciplinary staff is here to help you address your most challenging issues in your response to COVID-19. We have compiled free resources in the ECRI COVID-19 Resource Center. Please contact us for assistance and to learn more.
Return to the ECRI COVID-19 Resource Center
* All material in this COVID-19 Resource Center, https://www.ecri.org/coronavirus-covid-19-outbreak-preparedness-center/, is protected by copyright, and all rights are reserved under international and Pan-American copyright conventions. However, in light of current circumstances, due to the COVID-19 pandemic, ECRI will permit the ECRI content accessible from this COVID-19 Resource Center to be reproduced and shared, in whole or in part, for non-commercial purposes only. If you desire to reproduce or share content from third-party sources cited within this Center, you must first seek approval from that third-party source.